Nonprotein components of certain enzymes are called cofactors. If the
cofactor is organic, then it is called a coenzyme. Coenzymes are
relatively small molecules compared to the protein part of the enzyme
and many
of the coenzymes are derived from vitamins. The coenzymes make up a part of the active site, since without the coenzyme, the enzyme will not function.




of the coenzymes are derived from vitamins. The coenzymes make up a part of the active site, since without the coenzyme, the enzyme will not function.
Vitamin A: Beta-Carotene
Beta-carotene
is the molecule that gives carrots, sweet potatoes, squash, and other
yellow or orange vegetables their orange color. It is part of a family
of chemicals called the carotenoids, which are found in many fruit and
vegetables, as well as some animal products such as egg yolks.
Carotenoids were first isolated in the early 19th century, and have been
synthesized for use as food colorings since the 1950s. Biologically,
beta-carotene is most important as the precursor of vitamin A in the
human diet. It also has anti-oxidant properties and may help in
preventing cancer and other diseases.
Introduction
The long chain of alternating double
bonds (conjugated) is responsible for the orange color of beta-carotene.
The conjugated chain in carotenoids means that they absorb in the
visible region - green/blue part of the spectrum. So beta-carotene
appears orange, because the red/yellow colors are reflected back to us.

Vitamin A
Vitamin A has several functions in the
body. The most well known is its role in vision - hence carrots "make
you able to see in the dark". The retinol is oxidized to its aldehyde,
retinal, which complexes with a molecule in the eye called opsin. When a
photon of light hits the complex, the retinal changes from the 11-cis
form to the all-trans form, initiating a chain of events which results
in the transmission of an impulse up the optic nerve. A more detailed
explanation is in Photochemical Events.

Other roles of vitamin A are much less
well understood. It is known to be involved in the synthesis of certain
glycoproteins, and that deficiency leads to abnormal bone development,
disorders of the reproductive system, xerophthalmia (a drying condition
of the cornea of the eye) and ultimately death.
Vitamin A is required for healthy skin
and mucus membranes, and for night vision. Its absence from diet leads
to a loss in weight and failure of growth in young animals, to the eye
diseases; xerophthalmia, and night blindness, and to a general
susceptibility to infections. It is thought to help prevent the
development of cancer. Good sources of carotene, such as green
vegetables are good potential sources of vitamin A. Vitamin A is also
synthetically manufactured by extraction from fish-liver oil and by
synthesis from beta-ionone.
Vitamin A is structurally related to carotene. Carotene is converted into vitamin A in the liver. Two molecules of vitamin A are formed from on molecule of beta carotene.

Oxidation: If you
compare the two molecules, it is clear that vitamin A (retinol) is very
closely related to half of the beta-carotene molecule. One way in which
beta-carotene can be converted to vitamin A is to break it apart at the
center and is thought to be most important biologically. The breakdown
of beta-carotene occurs in the walls of the small intestine (intestinal
mucosa) and is catalyzed by the enzyme beta-carotene dioxygenase to form
retinal.
Reduction Reaction: The
retinal reduced to retinol by retinaldehyde reductase in the
intestines. This is the reduction of an aldehyde by the addition of
hydrogen atoms to make the alcohol, retinol.
Esterification Reaction:
The absorption of retinol from the alimentary tract is favored by the
simultaneous absorption of fat or oil, especially if these are
unsaturated. Retinol is esterified to palmitic acid and delivered to the
blood via chylomicrons. Finally the retinol formed is stored in the
liver as retinyl esters. This is why cod liver oil used to be taken as a
vitamin A supplement. It is also why you should never eat polar bear
liver if you run out of food in the Arctic; vitamin A is toxic in excess
and a modest portion of polar bear liver contains more than two years supply!
Beta-carotene, on the other hand, is a safe source of vitamin A. The
efficiency of conversion of beta-carotene to retinol depends on the
level in the diet. If you eat more beta-carotene, less is converted, and
the rest is stored in fat reserves in the body. So too much
beta-carotene can make you turn yellow, but will not kill you with
hypervitaminosis.
FAD - Flavin Adenine Dinucleotide
The structure shown on the left is for FAD and is similar to NAD+
in that it contains a vitamin-riboflavin, adenine, ribose, and
phosphates. As shown it is the diphosphate, but is also used as the
monophosphate (FMN).
Introduction
In the form of FMN it is involved in the first enzyme complex 1 of the electron transport chain.
A FMN (flavin adenine mononucleotide) as an oxidizing agent is used to
react with NADH for the second step in the electron transport chain. The
simplified reaction is:
NADH + H+ + FMN → FMNH2 + NAD+
Red.Ag. Ox.Ag.
Red.Ag. Ox.Ag.
Note the fact that the two hydrogens and 2e- are "passed along" from NADH to FFMN. Also note that NAD+
as a product is back to its original state as an oxidizing agent ready
to begin the cycle again. The FMN has now been converted to the reducing
agent and is the starting point for the third step.

Coenzyme Q or Ubiquinone
Ubiquinone: As its name
suggests, is very widely distributed in nature. There are some
differences in the length of the isoprene unit (in bracket on left) side
chain in various species. All the natural forms of CoQ are insoluble in
water, but soluble in membrane lipids where they function as a mobile
electron carrier in the electron transport chain. The long hydrocarbon chain gives the non-polar property to the molecule.
CoQ acts as a bridge between enzyme
complex 1 and 3 or between complex 2 and 3. Electrons are transferred
from NADH along with two hydrogens to the double bond oxygens in the
benzene ring. These in turn convert to alcohol groups. The electrons are
then passed along to the cytochromes in enzyme complex 3.

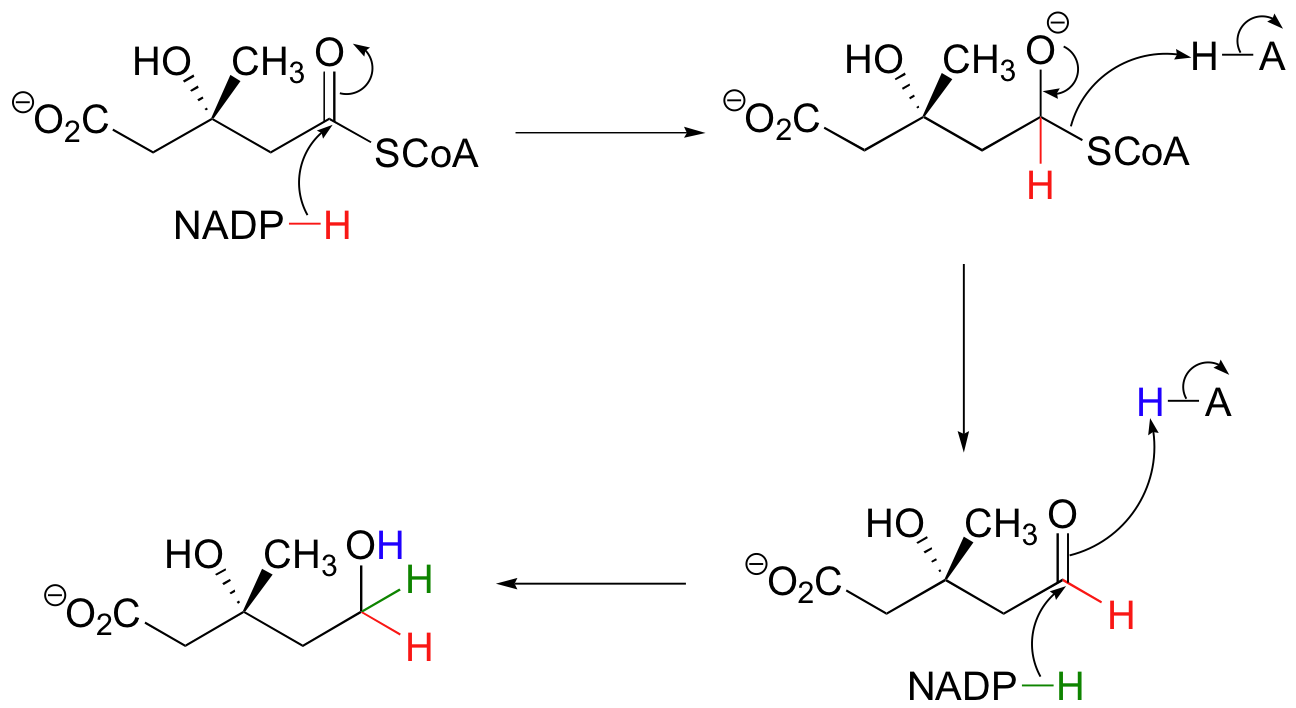
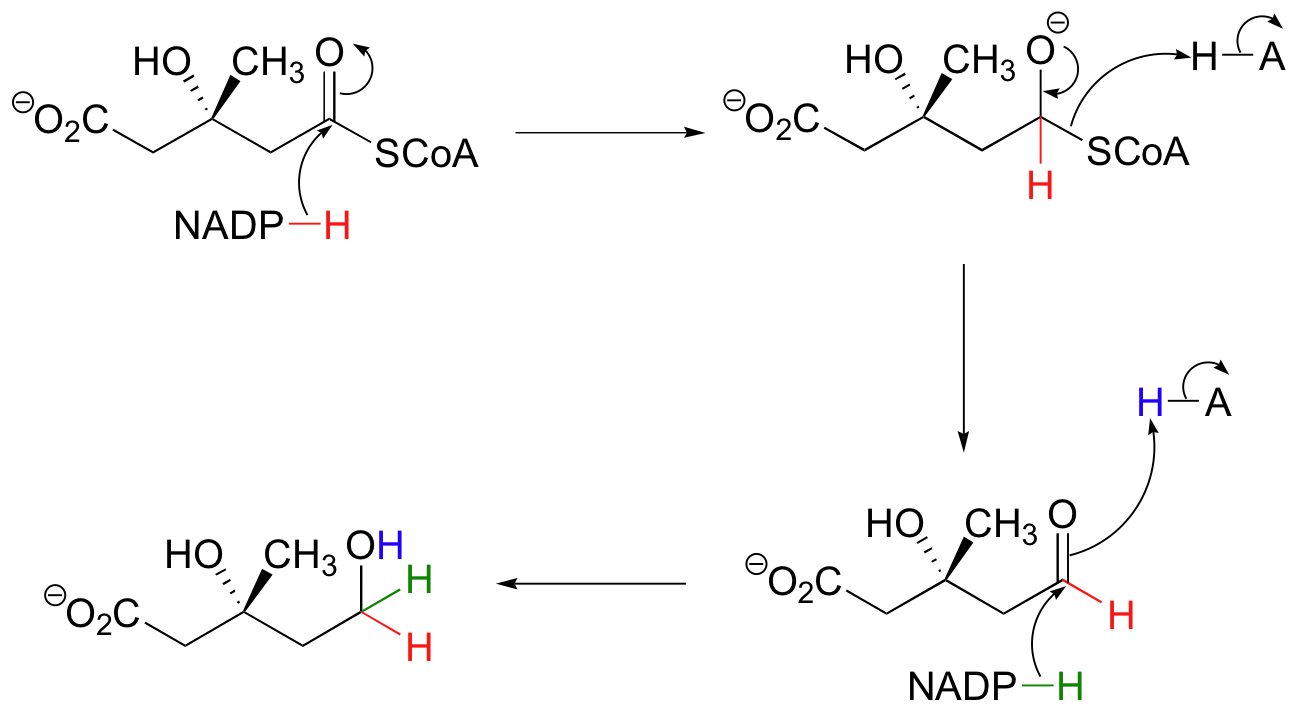
Coenzyme A
Although not used in the electron
transport chain, Coenzyme A is a major cofactor which is used to
transfer a two carbon unit commonly referred to as the acetyl group. The structure has many common features with NAD+
and FAD in that it has the diphosphate, ribose, and adenine. In
addition it has a vitamin called pantothenic acid, and finally
terminated by a thiol group. The thiol (-SH) is the sulfur analog of an
alcohol (-OH). The acetyl group (CH3C=O) is attached to the
sulfur of the CoA through a thiol ester type bond. Acetyl CoA is
important in the breakdown of fatty acids and is a starting point in the
citric acid cycle.
Nicotinamide Adenine Dinucleotide
In the graphic below, the structure for the coenzyme, NAD+, Nicotinamide Adenine Dinucleotide is shown. Nicotinamide is from the niacin vitamin. The NAD+
coenzyme is involved with many types of oxidation reactions where
alcohols are converted to ketones or aldehydes. It is also involved in
the first enzyme complex 1 of the electron transport chain.
Role of NAD+
One role of NAD+ is to initiate the electron transport chain by the reaction with an organic metabolite
(intermediate in metabolic reactions). This is an oxidation reaction
where 2 hydrogen atoms (or 2 hydrogen ions and 2 electrons) are removed
from the organic metabolite. (The organic metabolites are usually from
the citric acid cycle and the oxidation of fatty acids--details in
following pages.) The reaction can be represented simply where M = any
metabolite.
MH2 + NAD+ → NADH + H+ + M: + energy
One hydrogen is removed with 2 electrons as a hydride ion (H-) while the other is removed as the positive ion (H+). Usually the metabolite is some type of alcohol which is oxidized to a ketone.


Alcohol Dehydrogenase
The NAD+ is represented as
cyan in belowthe graphic. The alcohol is represented by the space
filling red, gray, and white atoms. The reaction is to convert the
alcohol, ethanol, into ethanal, an aldehyde.
CH3CH2OH + NAD+ → CH3CH=O + NADH + H+
This is an oxidation reaction and results in the removal of two hydrogen ions and two electrons which are added to the NAD+, converting it to NADH and H+.
This is the first reaction in the metabolism of alcohol. The active
site of ADH has two binding regions. The coenzyme binding site, where
NAD+ binds, and the substrate binding site, where the alcohol binds. Most of the binding site for the NAD+
is hydrophobic as represented in green. Three key amino acids involved
in the catalytic oxidation of alcohols to aldehydes and ketones. They
are ser-48, phe 140, and phe 93.

Cobalamin
Cobalamin, or Vitamin B12,
is the largest and the most complex out of all the types of Vitamins.
The discovery of Cobalamin was made as scientists were seeking to find a
cure for pernicious anemia, an anemic disease caused by an absence of
intrinsic factor in the stomach. Cobalamin was studied, purified, and
collected into small red crystals, and its crystallize structure was
determined during an X ray analysis experiment conducted by Scientist
Hodkin. A molecule structure of Cobalamin is simple, yet contains a lot
of different varieties and complexes as shown in Figure 1. The
examination of the vitamin’s molecular structure helps scientists to
have a better understanding of how the body utilizes Vitamin B12 into building red blood cells and preventing pernicious anemia syndromes.

Figure 1
The metalloenzyme structure of Cobalamin
presents a corrin ring with Cobalt, the only metal in the molecule,
positioned right in the center of the structure by four coordinated
bonds of nitrogen from four pyrrole groups. These four subunit groups
are separated evenly on the same plane, directly across from each other.
They are also connected to each other by a C-CH3 methylene
link on the other sides, by a C-H on one side and by two pyrroles
directly coming together. Together, they form a perfect corrin ring as
shown in figure 2. The fifth ligand connected to Cobalt is a nitrogen
coming from the 5,6-dimethhylbenzimidazole. It presents itself as an
axial running straight down from the cobalt right under the corrin ring.
This benzimidazole is also connected to a five carbon sugar, which
eventually attaches itself to a phosphate group, and then straps back to
the rest of the structure. Since the axial is stretched all the way
down, the bonding between the Cobalt and the 5,6-dimethylbenzimidazole
is weak and can sometimes be replaced by related molecules such as a
5-hydrozyl-benzimidazole, an adenine, or any other similar group. In the
sixth position above the Corrin ring, the active site of Cobalt can
directly connect to several different types of ligands. It can connect
to CN to form a Cyanocobalami, to a Methyl group to form a
methylcobalamin, to a 5’-deoxy adenosy group to form an
adenosylcobalamin, and OH, Hydroxycobalamin. Cobalt is always ready to
oxidize from 1+ change into 2+ and 3+ in order to match up with these R
groups that are connected to it. For example, Hydroxocobalamin contains
cobalt that has a 3+ charge while Methyladenosyl contains a cobalt that has a 1+ Charge.

Figure 2
The point group configuration of Cobalamin is C4v.
In order to determine this symmetry, one must see that the structure is
able to rotate itself four times and will eventually arrive back to its
original position. Furthermore, there are no sigma h plane and no
perpendicular C2 axe. However, since there are sigma v planes
that cut the molecules into even parts, it is clear to determine that
the structure of Cobalamin is a C4v. With Cobalt being the
center metal of the molecule, Cobalamin carried a distorted octahedral
configuration. The axial that connects Cobalt to the 5,6 dimethyl
benzimidazole is stretched all the way down to the bottom. Its distance
is several times longer than the distance from the Cobalt and the
attached R group above it. This sometimes can also be referred to as a
tetragonal structure. The whole shape overall is similar to an
octahedral, but the two axial groups are different and separated into
uneven distances. Since there is only one metalloenzyme center in the
system, the point group and configuration just mentioned is also
assigned to the structure as a whole. Since the metallocoenzyme
structure is stretched out, it is quite weakly coordinated and can be
break apart or replaced with other groups as mentioned above.
Scientists have shown that both IR and
Raman Spectroscopy were used to determine the structure of the molecule.
This is determined by observing the character tables of point group C4v,
the point group symmetry of Cobalamin. On the IR side, one can see that
there are groups such as drz, (x, y), (rz, ry). On the other hand, on
the Raman side, there are groups such as x square +y square, z square, x
square – y square, xy, xz, yz. The Raman side indicated that there were
stretching modes in the molecule and relates back to the stretching of
the 5,6 dimethyl benzimidazole axial that connected directly below the
Cobalt metal. The stretching can be seen in Figure 3.

Figure 3
Cobalamin enzymes can catalyze a few
different types of reactions. One of them is the reaction of
Intramolecular rearrangements. During this rearrangement coenzyme is
exchanged to the two groups attached to adjacent carbon atoms. Another
reaction involves transferring the methyl group in certain methylation
reactions, such as the conversion of homocysteine to methionine,
biosysnthesis of choline and thymine etc. These interactions can bring
beneficial values to the biological bodies.
Cobalamin has many beneficial effects in
regard to biological existences. They play a role to maintain healthy
body system and help to aid the production of the body’s genetic
materials. Cyanocobalamin, one type of cobalamin, works to generate the
forming of red blood cells and heal many different damages in the
nervous system. Cobalamin also serves as a vital role in the metabolism
of fatty acids essential for the maintainence of myelin. Studies have
shown that people with Vitamin B12 deficiency will reveal
irregular destruction of the myeline shealth, which leads to parlysis
and death. Some of the other symptoms of the lack of cobalamin are poor
growth, megaloblastic bone marrow, Gi tract changes, Leucoopenia and
hyper-segmented nutrophills, degenerative changes in spinal cord and
nervous system and excretion of methyl malonic acid and homocystin in
urine.
Throughout the years, Vitamin B12 has
shown to be essential for the functioning of the nervous system and the
production of red blood cell. A study conducted by researchers at the
National Institutes of Health, Trinity College Dublin, suggested that a
deficiency in Vitamin B12 might increase the risk of neural
tubes defect in children (Miller). Therefore, by studying the structure
and function of Cobalamin, scientists can experiment and form Vitamin
B12 in their laboratories and serve the community as a whole.
1 comment:
I started on COPD Herbal treatment from Ultimate Life Clinic, the treatment worked incredibly for my lungs condition. I used the herbal treatment for almost 4 months, it reversed my COPD. My severe shortness of breath, dry cough, chest tightness gradually disappeared. Reach Ultimate Life Clinic via their website www.ultimatelifeclinic.com . I can breath much better and It feels comfortable!
Post a Comment